Mima Clinical
Continuous and Adaptive PRO Collection for Clinical Trials
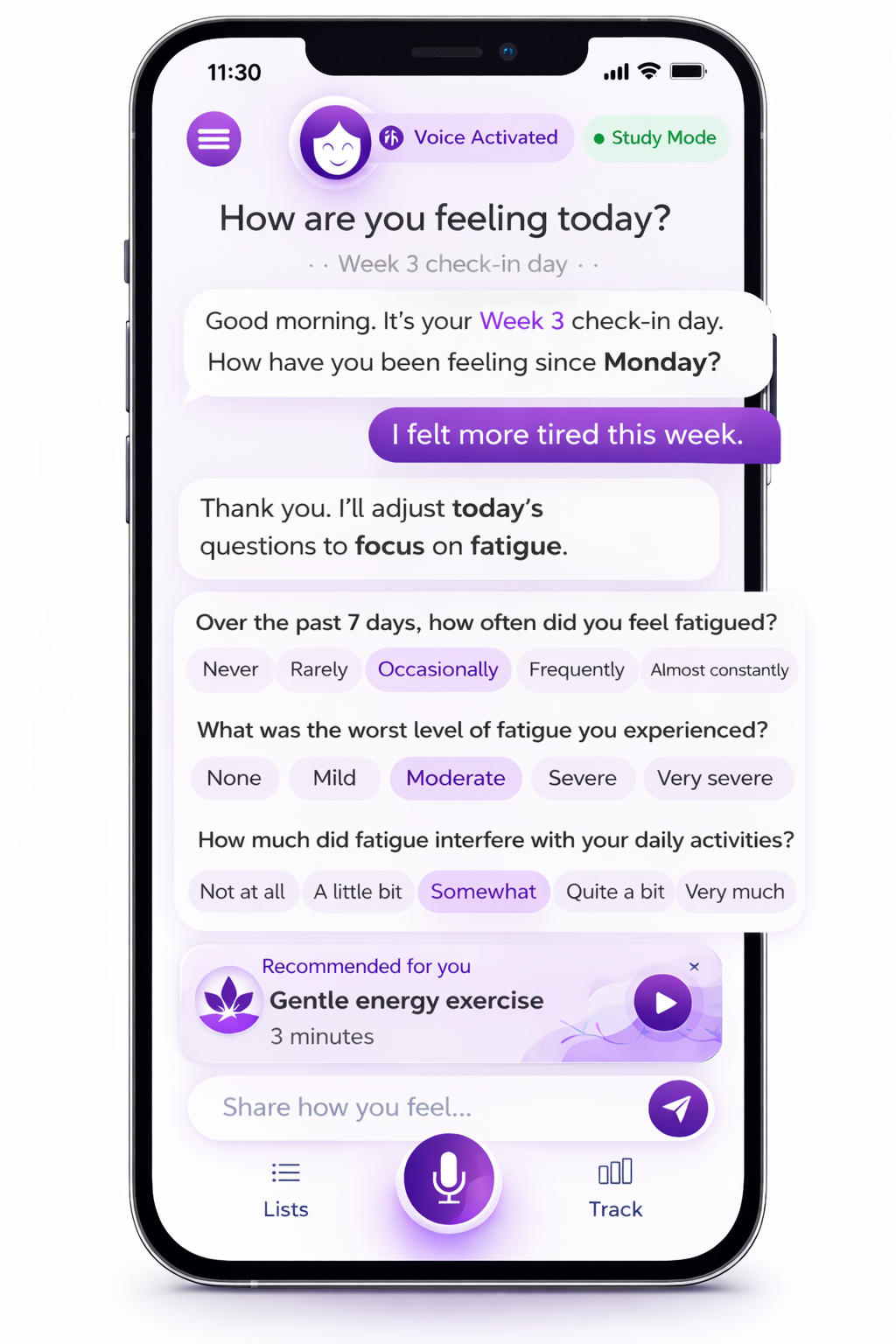
Mima Clinical captures patient-reported symptoms in real time, adapts follow-up questions to what patients report, and helps surface signals that emerge beyond predefined questionnaires and predefined timepoints.
Continuous, adaptive follow-up
Captures non-predefined symptoms
Structured exports for study workflows
Real-time cohort visibility
Estimated CTCAE-aligned severity mapping (configurable)
Patient-reported symptoms can be mapped to an estimated CTCAE-aligned severity signal to support site workflows and structured reporting.
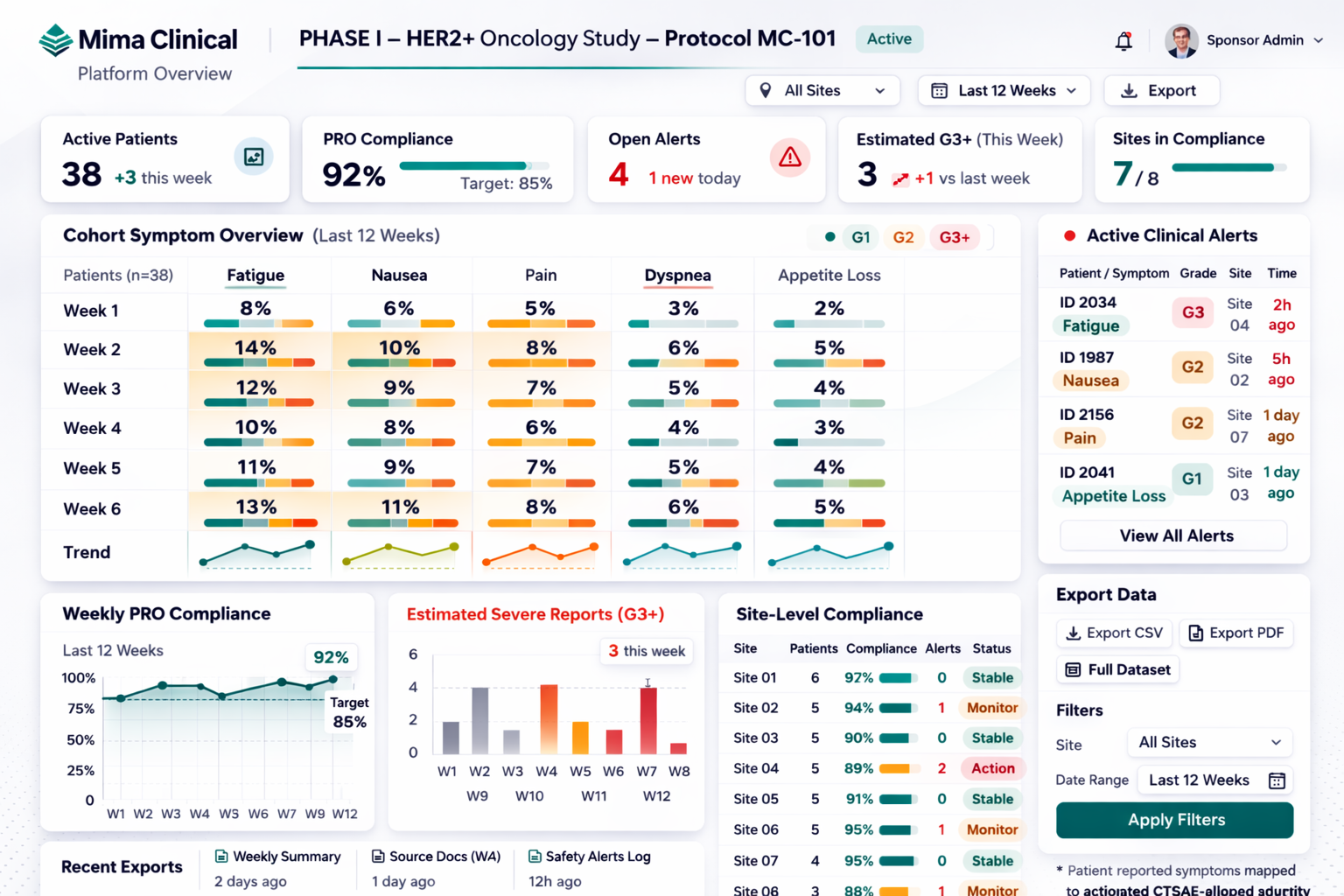
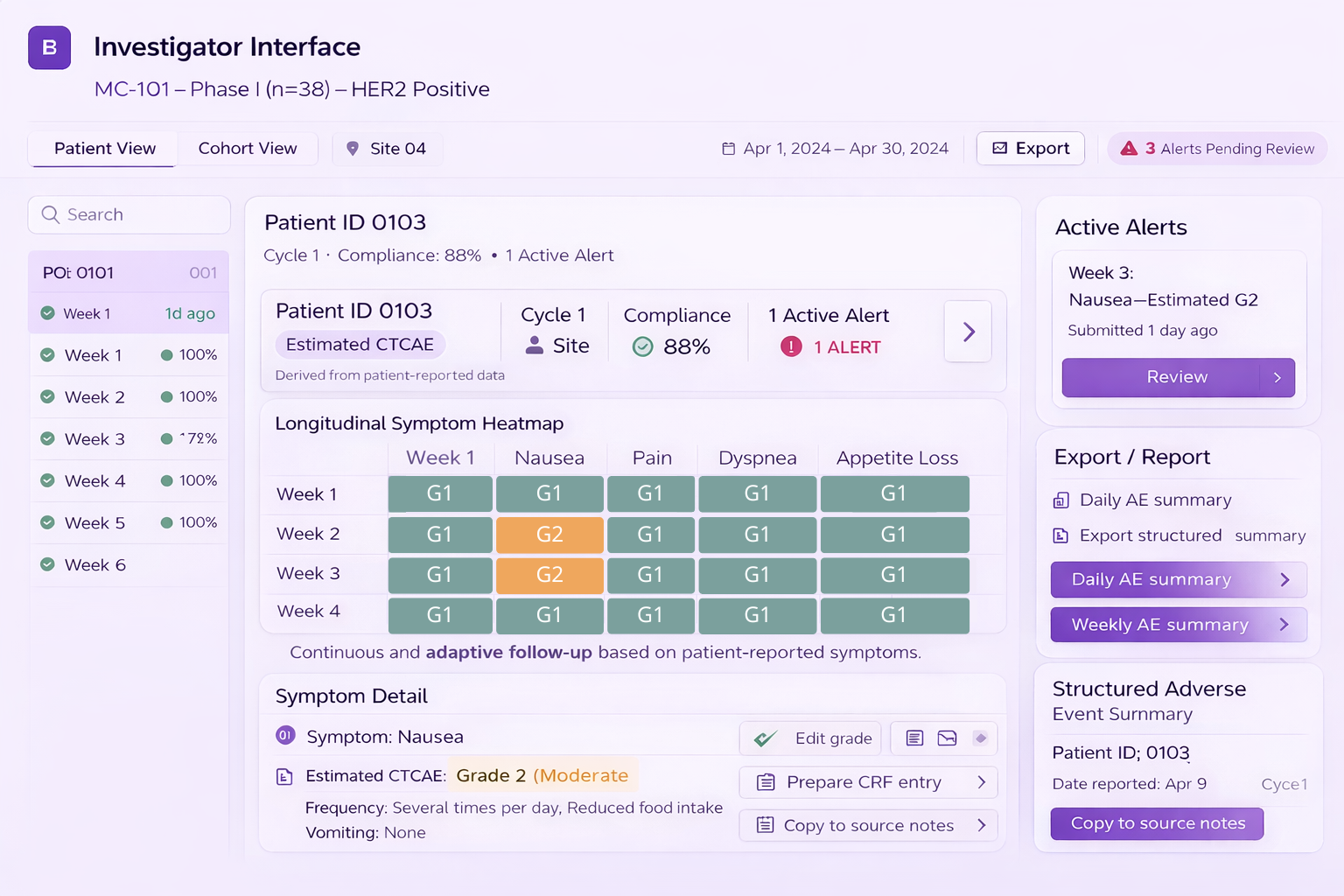
Dashboard example shown for illustration. Patient-level data can be configured for anonymized display depending on study needs.
Limitations of Traditional ePRO Systems
Many trial ePRO approaches are constrained by predefined timepoints and predefined AE lists, making it harder to capture emerging, patient-initiated signals during the study.
Predefined Timepoints
Reporting is often limited to scheduled visits and fixed intervals, reducing visibility between windows.
Preselected AE Lists
Collection frequently focuses on predefined symptoms only, which can miss signals that emerge during treatment.
Standalone Research Tools
Research-only applications often face lower long-term engagement, impacting sustained compliance.
Mima Clinical Architecture
A patient-centered platform with a dedicated research layer for patients, investigators, and sponsors. Follow-up adapts to reported symptoms, enabling continuous collection alongside protocol-aligned questionnaires.
Patient Interface
- Study-specific onboarding and consent flow
- PRO-CTCAE aligned questionnaires
- Protocol reporting intervals (J1, J8, weekly)
- Continuous and adaptive follow-up based on patient-reported symptoms
- Study overview and site contact access
Symptom grading displayed in-app can be configured as patient-reported grading aligned with PRO-CTCAE, and as estimated CTCAE-aligned severity mapping when used to support study workflows.
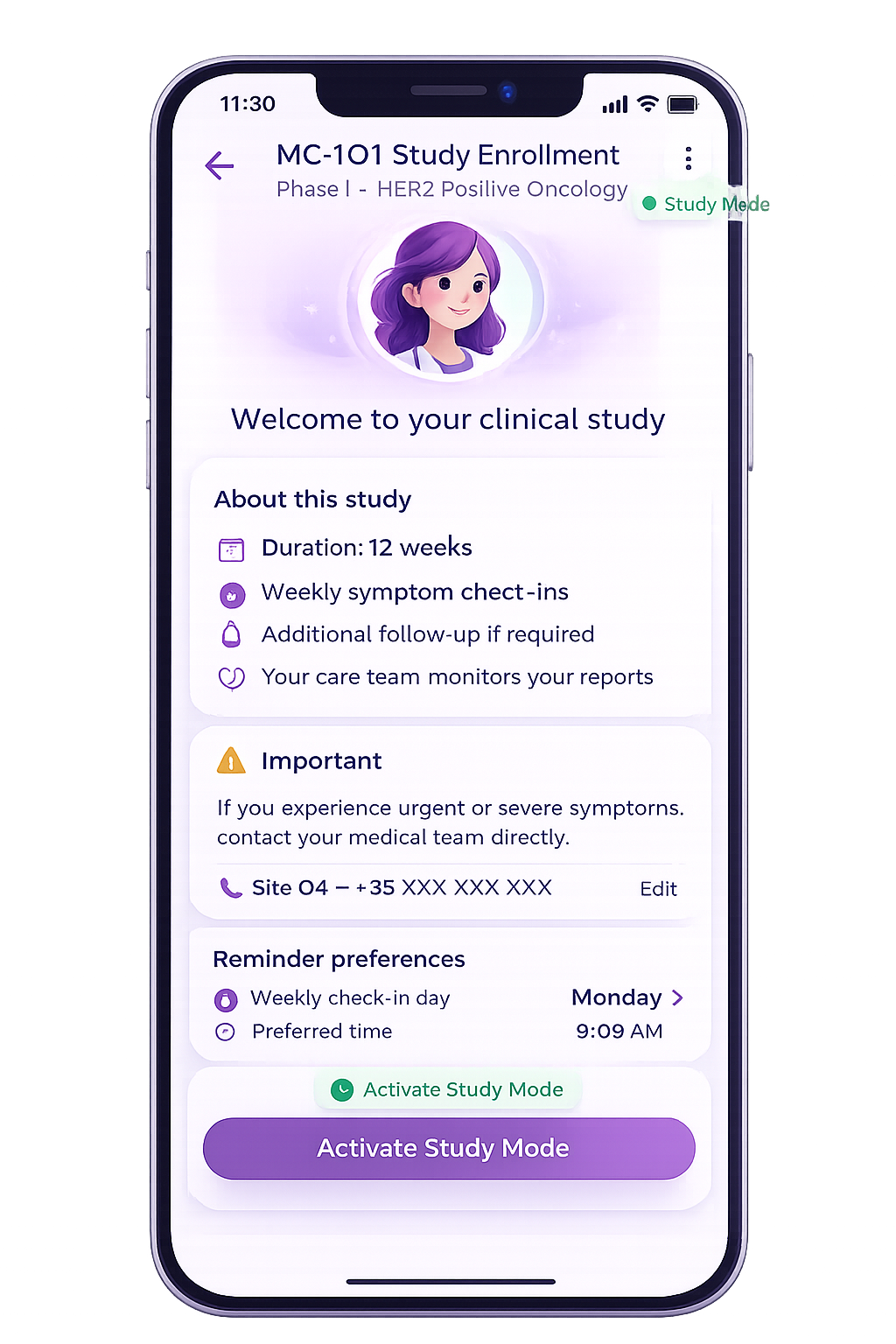
Patient Study Onboarding
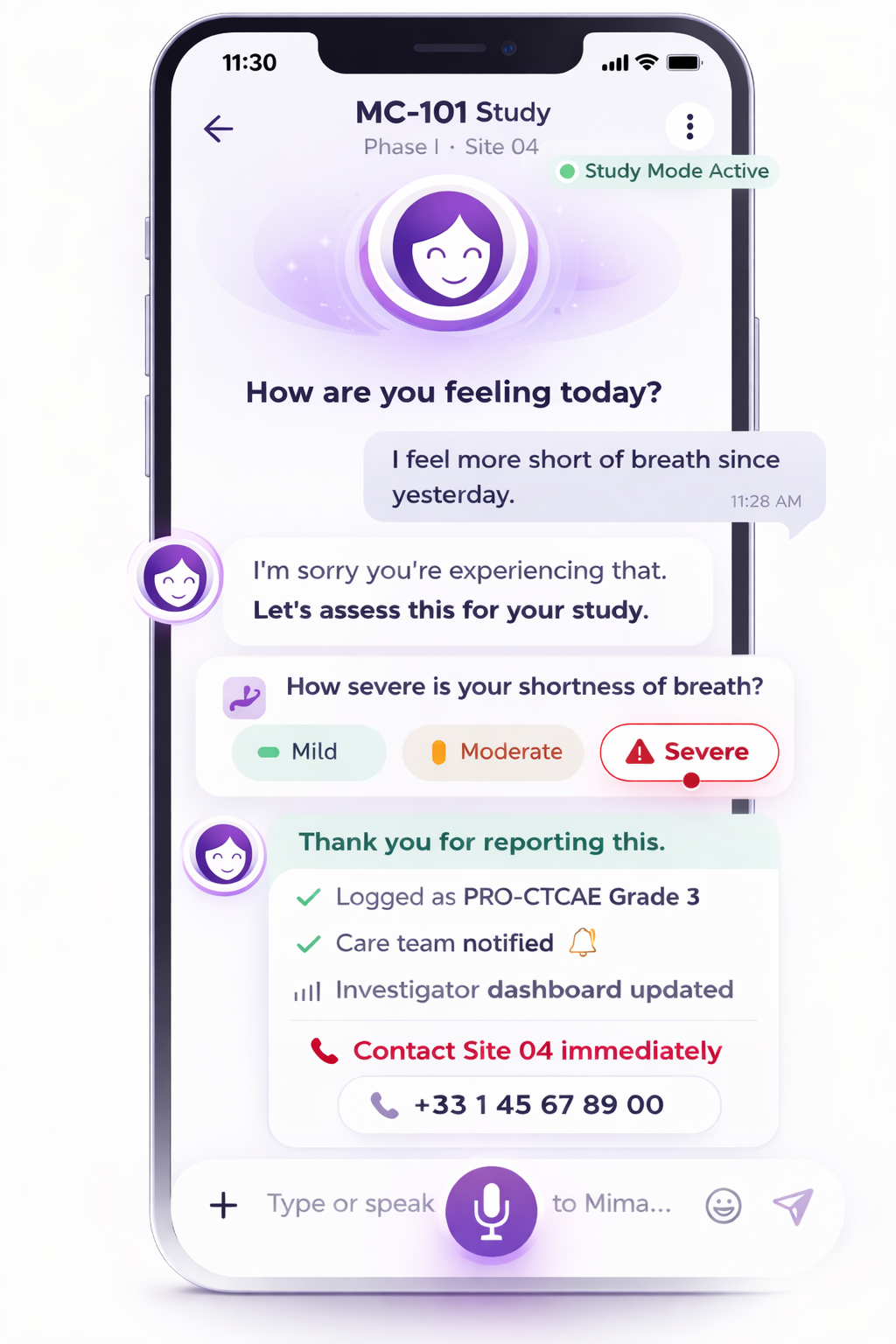
Adaptive PRO Check-in
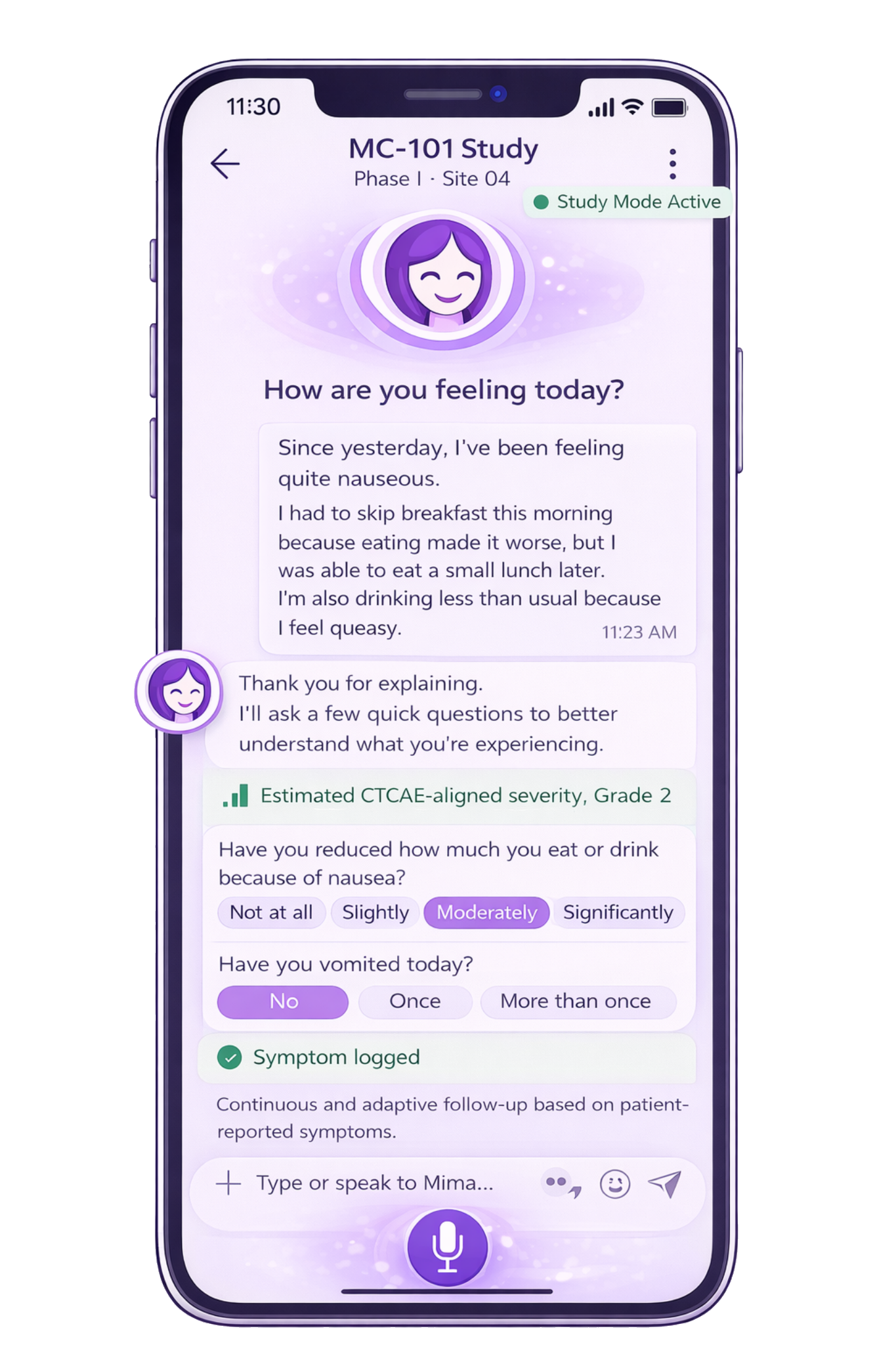
Patient-initiated AE & Adaptive Grading
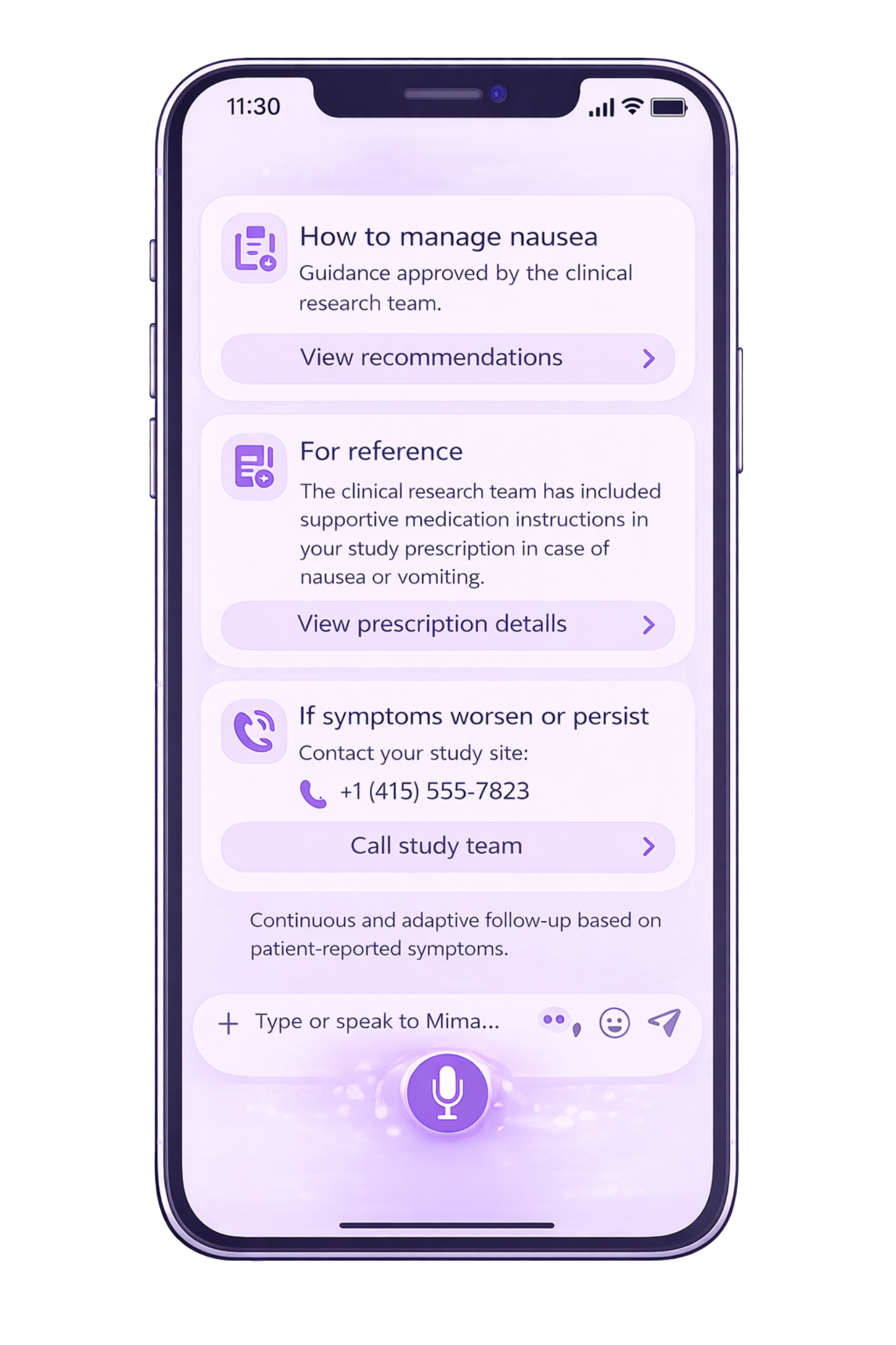
Guidance, Prescription & Escalation
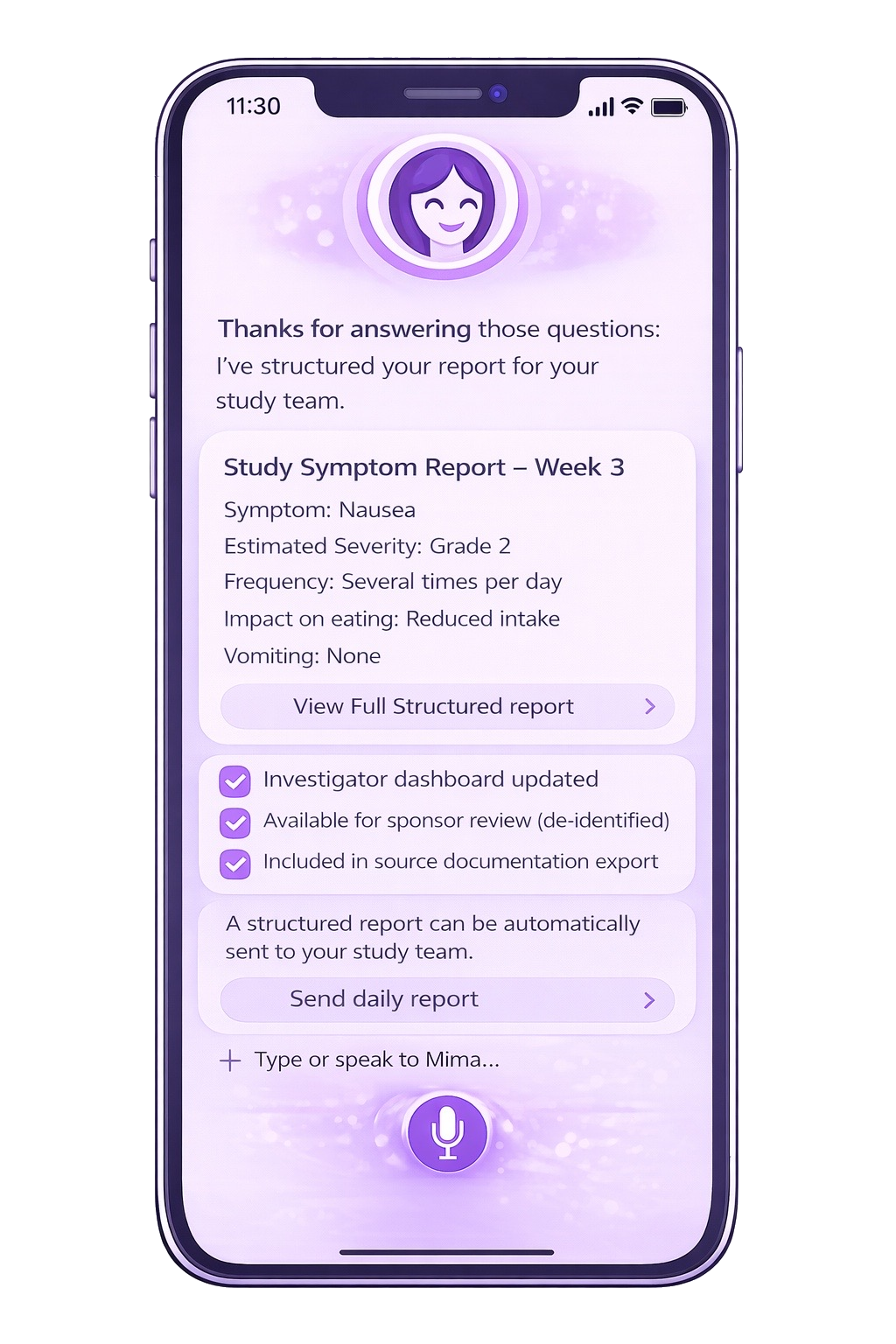
Structured AE Summary for Site Workflow
Investigator Interface
- Longitudinal symptom tracking per patient (ID-based)
- Heatmap overview for cohort monitoring
- Compliance monitoring and follow-up status
- Threshold-based alerts aligned with protocol rules
- Structured exports for source documentation and CRF preparation
Source documentation summaries
Daily or weekly summaries of patient-reported AEs and free-text can be generated for study coordinators and site workflows.
Sponsor Interface
- Cohort-level visibility across sites
- Compliance and reporting cadence metrics
- Aggregated symptom trends and signal tracking
- Site-level monitoring views
- Exportable structured datasets for analysis
Sponsor views can be configured for anonymized patient identifiers and site-level aggregation depending on study requirements.

Real-Time Signal Routing
Routing rules are configured at study level. Patient reports can trigger adaptive follow-up and protocol-aligned escalation.
Patient Report
Adaptive Follow-up and Threshold Rules
Site Notification and Study Workflow
Sponsor Visibility (Configured)
Mima Clinical supports continuous capture and structured reporting of patient-reported outcomes, including protocol-aligned questionnaires, adaptive symptom follow-up, and configurable study routing.
Sustained Patient Compliance
Mima Clinical operates inside a broader patient companion environment, supporting sustained engagement beyond scheduled questionnaires.
- Embedded within patient daily-use environment
- Continuous interaction beyond scheduled questionnaires
- Reduced app fatigue through conversational check-ins and lightweight inputs
Example Workflow • Phase I Oncology Study (n=38)
Example flow illustrating continuous patient-initiated reporting alongside protocol check-ins, adaptive follow-up, and structured study reporting.
Study onboarding and baseline assessment
Protocol check-ins (weekly / scheduled)
Patient-initiated symptom reporting at any time
Adaptive follow-up questions based on reported symptoms
Site workflow and study documentation (summaries / exports)
Sponsor oversight through configured dashboards
Comparison Snapshot
A practical view of differences often discussed by sites and study teams.
| Capability | Mima Clinical | Standard PRO Apps |
|---|---|---|
| Continuous patient-initiated reporting | Yes, inside the companion chat | Varies by deployment |
| Adaptive follow-up based on symptoms | Yes, dynamic question routing | Typically predefined & fixed intervals |
| Captures non-predefined symptoms | Yes, beyond predefined AE lists | Typically predefined & fixed intervals |
| PRO-CTCAE aligned questionnaires | Yes | Varies by deployment |
| Estimated CTCAE-aligned severity mapping | Yes • algorithmic severity estimation from patient-reported symptoms (configurable) | Typically requires investigator grading; no automated severity estimation |
| Site documentation support (summaries/exports) | Daily/weekly source documentation summaries | Varies by deployment |
| Sponsor dashboard visibility | Configured, cohort-level views | Varies by deployment |
Compliance and Data Governance
Study-Level Integration
Mima Clinical is configured at study level with protocol-specific questionnaires, routing rules, dashboard access, and structured site onboarding.
- Study configuration
- Questionnaire customization
- Dashboard activation
- Site onboarding
- Patient activation via study code
Discuss Mima Clinical for your next study
We support Phase I to Phase III and post-approval studies, as well as multi-site trials, with continuous, adaptive patient-reported outcomes and structured study reporting.
Contact our team